STOP 301: Open-label Safety and Tolerability of Chronic Intermittent Usage for 24/52 Weeks of INP104 [Nasal Dihydroergotamine Mesylate (DHE) Administered by Precision Olfactory Delivery (POD®) Device] in Migraine Headache
posted in Presentations by dharmendra.asimi@trndigital.com
Poster presented at AHS 2019 Annual Meeting, poster #P44.
Poster Presenter: Stephen B. Shrewsbury, MD
Non-presenting Authors: Maria Jeleva, PhD; Meghan Swardstrom, BSc
Background
Migraine is a common, disabling disorder. IV DHE is an effective abortive therapy; however, the nasal formulation, Migranal®, has poor bioavailability and variable efficacy. We found INP104 (1.45 mg DHE) to have 4X greater systemic exposure than Migranal (2 mg) as a result of delivery technology4. In INP104, the POD system keeps drug and HFA propellant separate until actuation then propels a focused drug-containing stream to the olfactory epithelium of the upper nasal space where rapid absorption into the systemic circulation occurs. No product has previously utilized this space for systemic drug delivery so the STOP 301 study was designed to assess the safety of chronic, repetitive administration of DHE on olfactory epithelium integrity and function.
Methods
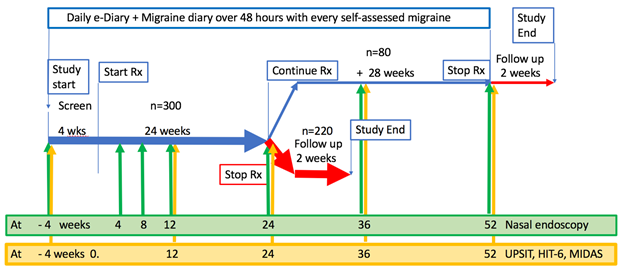
Approximately 300 patients will enter the 24week study, allowing ³150 to be exposed to INP104 ³2x/month x 24 weeks, with ~80 enrolling into a further 28week treatment period so ³50 subjects will complete 52 weeks of exposure. Main inclusion criteria: 18 to 65 years; migraine with or without aura, ³2 attacks/month for previous 6 months; >80% e-dairy compliance; contracepting. Main exclusion criteria: trigeminal autonomic cephalalgias; migraine aura without headache, hemiplegic migraine or migraine with brainstem aura; chronic migraines, medication overuse headache or other chronic headache syndromes (³15 headache days in screening); status migrainosus; positive HIV, HBsAg or HCV antibodies; CAD, symptoms or findings consistent with coronary artery vasospasm, including Prinzmetal’s variant angina; significant risk factors for CAD, current tobacco user, smoking history (³10 cigarettes/day within the last 12 months), history of diabetes, peripheral arterial disease, Raynaud’s phenomenon; potentially unrecognized CAD on history, examination, or ECG; abnormal, clinically significant laboratory tests at screening; recent acute illness or uncontrolled infection; recurrent sinusitis or epistaxis, or CRSwNP; significant nasal congestion, physical blockage in either nostril, significantly deviated nasal septum, septal perforation, or any pre-existing upper nasal mucosal abnormality; triptan or ergot use > 12 days/month in the 2 months prior or during screening; use of barbiturates/barbiturate containing compounds or opiates > 7 days/month or unstable usage pattern in 2 months prior or during screening.
All subjects will have extensive migraine history, healthcare utilization and quality of life assessments conducted during screening, on treatment, and following all exposures to INP104 (for self-assessed migraine) recorded in a migraine efficacy e-diary. Olfactory mucosal integrity and function will be collected by endoscopy of the upper (and lower) nasal spaces and by University of Pennsylvania Smell Identification Test (UPSIT) at intervals throughout exposure.
Results
The resulting study design is shown in the figure:
Conclusion
STOP 301 is designed to assess safety, tolerability and explore efficacy of repeat administration of DHE drug to the upper nasal space.
